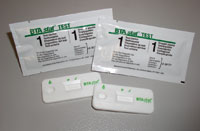
The BTA stat® test is a point of care technology for the early detection of recurrent bladder cancer. This method uses monoclonal antibodies to detect the presence of bladder tumor associated antigen in urine. It is a single-step, rapid immunochromatographic assay for bladder tumor-associated antigen in voided urine. The specificity of the BTA stat® test was 93 - 95% in patients with non-genitourinary diseases and cancers and healthy individuals tested as part of a multi-center study. The test has a sensitivity that is considerably higher than voided urine cytology, enabling detection of recurrent early stage and grade cancers that cytology often misses. Requiring three drops of urine, the result is delivered in only five minutes. The appearance of a line in the patient window indicates a positive result. The BTA stat® test requires one voided urine sample with no sample preparation. The BTA stat® test is CLIA waived and also available for prescription home use.
- Product Overview
-
- Educational Resources
-
- Videos
-