Point-of-care Testing: Facilitating Positive Outcomes in Chronic Disease Management
Wednesday, May 18, 2022
by Susan Garramone, Senior Clinical Marketing Manager
Siemens Healthcare Diagnostics Inc.
Introduction
Chronic diseases are the leading causes of death and disability in the U.S. and are leading drivers of the nation’s rising healthcare costs.1 Chronic diseases are defined as conditions that last 1 year or more and require ongoing medical attention, limit activities of daily living, or both. Heart disease, cancer, chronic lung disease, stroke, Alzheimer’s disease, diabetes, and chronic kidney disease are among the most common. A staggering 40% of adults in the U.S. have two or more chronic diseases.1 Unfortunately, chronic diseases are common, serious, and costly.
Common Comorbidities: Diabetes and Chronic Kidney Disease
Diabetes is the seventh-leading cause of death in the United States, affecting more than 37 million Americans.2,3 It is attributed to an estimated $327 billion in annual medical and lost productivity costs.2,3 Generally, care for patients with diabetes accounts for 1 in 4 healthcare dollars spent in the U.S., and a large portion of these costs results from diabetes-associated comorbidities that include potential neurological, cardiovascular, renal, and other chronic complications.3 In fact, diabetes is a risk factor for the development of chronic kidney disease (CKD); 1 in 3 adults with diabetes also have chronic kidney disease!4 In the U.S., where kidney disease is the tenth-leading cause of death, annual Medicare costs related to CKD are greater than $81 billion.5,6

In addition to major healthcare cost implications, complications from diabetes and CKD may have significant impact on patient well-being and quality of life. Early recognition and management of diabetes and CKD enable timely intervention and greater opportunity to protect patient health. Tragically, 40% of patients with diabetes and 90% of patients with CKD are unaware that they have these conditions.5,7

With primary care being the first point of contact and foundation of a person’s healthcare team, the ability to test, diagnose, consult, and manage diabetes and CKD in the primary care setting may provide the opportunity to prevent clinically worsening disease and improve patient outcomes and overall population health.8,9
Diabetes and Hemoglobin A1c Testing
Glycated hemoglobin (A1c, hemoglobin A1c, HbA1c), which reflects average levels of blood glucose over the previous 2–3 months, is the most widely used test to monitor chronic glycemic control and the efficacy of treatment.10 In contrast to blood glucose levels, which fluctuate throughout the day based on diet and activity, the HbA1c level reflects a patient’s glycemic control for the past 3 months.10
The American Diabetes Association (ADA) recommends the assessment of glycemic status (A1c or other glycemic measurement) at least two times per year in patients who are meeting treatment goals (and who have stable glycemic control) and at least quarterly, and as needed, in patients whose therapy has recently changed and/or who are not meeting glycemic goals.11 HbA1c testing can be performed in point-of-care (POC) settings such as a physician’s office or laboratory, though the ADA states that POC testing for A1c provides opportunity for more timely treatment changes.11
According to the CDC, between 2011 and 2016, only 67.3–71.4% of U.S. adults with diabetes reported following the recommended testing guidelines.12 This statistic is very concerning from a population health perspective, as low adherence to HbA1c testing has been shown to lead to higher HbA1c levels, which in turn may lead to complications and comorbidities.13 Research shows that every 1% decrease in the HbA1c level in a diabetes patient can remarkably lower the risk of complications, underscoring the importance of HbA1c monitoring and control.14

A review of the literature suggests that incorporating A1c testing into the patient visit and customizing the consultation to the patient’s status appear to help physicians influence their patients to improve their glycemic control.15
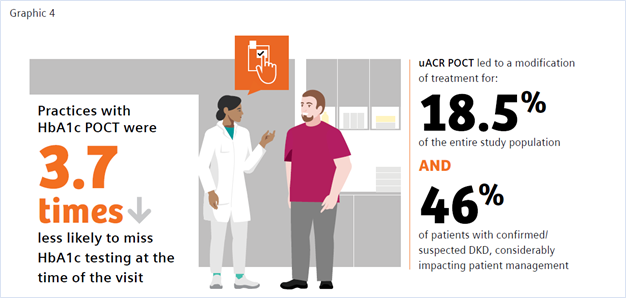
The benefit of POC HbA1c testing has been widely reported in the literature.9,15-19 Of note is a study by Crocker et al. that demonstrated that POC HbA1c testing led to 3.7 times decreased likelihood of missing HbA1c testing.9 Nearly 1 in 4 of the individuals tested were found to have clinically worsening diabetes. Their diagnosis using POC testing enabled more timely intervention in diabetes management. A recent review by Rhyu et al. also highlighted the benefits of POC HbA1c testing in the physician’s office, where the authors concluded that “POC A1c testing in primary care, if widely available and integrated into workflow, has the potential to positively impact diabetes care. Real-time feedback may change patient and physician behaviors, allowing earlier therapeutic intensification.”15
In addition to addressing the clinical benefit, numerous studies have shown the cost-effectiveness of implementing POC HbA1c testing.16-20 Rosa et al. found that “compared to a centralized laboratory test, the use of the POC-A1c device in a healthcare unit increased the chance of the early control of type 2 diabetes and reduced costs in relation to DM-related outcomes.”19 Crocker et al. reported that point-of-care HbA1c testing “can significantly improve clinical operations with cost reductions through improved practice efficiency,” realizing a potential savings from improved efficiency of $24.64 per patient. This savings resulted from 89% and 85% decreases in follow-up phone calls and letters, respectively, and a 61% decrease in patient revisits.17
CKD and Urinary Albumin-to-Creatinine Ratio (uACR)
The benefits of uACR testing at the point of care have similarly been studied, with findings of accuracy, convenience, improved patient access, and improvements in test adherence.21-25 The ADA recommends performing a spot uACR and estimated glomerular filtration rate (eGFR) test at least annually in patients with type 1 diabetes with duration of >5 years and in all patients with type 2 diabetes regardless of treatment. Patients with diabetes and urinary albumin >300 mg/g creatinine and/or an estimated glomerular filtration rate of 30–60 mL/min/1.73 m2 should be monitored twice annually to guide therapy.26
A recent study by Christofides and Desai discussed the importance of monitoring albuminuria in patients with type 2 diabetes as an essential tool in the detection of onset of CKD and monitoring disease progression. They stated, “Access to UACR testing may be improved by using Clinical Laboratory Improvements Amendments (CLIA)-waived point of care UACR testing options, that is, those approved for use closer to the patient and not necessarily in a central laboratory,” and noted that using a CLIA-waived POC uACR option was a means to optimize test ordering.21
CLIA stands for Clinical Laboratory Improvement Amendments of 1988, a set of U.S. regulatory standards that apply to all clinical laboratory testing performed on humans (except for clinical trials and basic research), regardless of where the test is performed. A test system or product that is granted waived status is defined, in part, as a methodology that is simple and accurate to render negligible the likelihood of erroneous results.27 Many POC tests meet this classification, as they require no formal training, and operators must only follow the manufacturer’s instructions for use. Waived POC tests are portable, easy to use, and offer accurate, quick near-patient results, making them particularly useful for testing in a physician’s office. In this environment a physician can test a patient at the time of visit and obtain the result in real time, providing the opportunity to counsel the patient and make treatment adjustments at the time of the office visit.28
There is a documented need for strategies to improve routine CKD assessment nationwide and overcome testing barriers to increase test adherence.
A study by Folkherts et al. reported that physicians treating patients with diabetes are selectively adhering to chronic kidney disease screening guidelines where, despite recommendations to monitor both eGFR and urinary ACR, less than half of patients were screened for albuminuria during the 1-year follow-up.29 Additionally, a large retrospective study of patients at-risk for CKD by Alfego et al. found only 21% of adults at risk for CKD and 32.2% of patients with diabetes specifically had guideline-recommended uACR testing.30 Schultes et al. found that the implementation of ACR POCT in daily general practice can improve the diagnosis of diabetic kidney disease (DKD) in diabetes, which may support improved management of CKD.22 DKD was newly diagnosed in 8.6% of the entire study population and DKD was suspected in 9.9% based on their ACR POCT values. In 18.5% of the entire study population ACR POCT led to a change in medication, demonstrating a benefit in patient management. When surveyed regarding the relevance of the ACR POCT, 75% of physicians considered the test very important for people with diabetes; 25% rated it important.
Conclusion
Primary care physicians treat at least 90% of patients with diabetes in the United States.31 Patients with diabetes are at risk of developing CKD and monitoring for both conditions is important for disease management. Guidelines call for routine testing of HbA1c and uARC for disease monitoring and management.12,27 Adoption of in-office POC HbA1c and uACR testing may provide physicians an important tool toward the goal of closing the diabetes care gap, enabling convenient, efficient, cost-effective testing, and facilitating improved patient adherence to testing. Increased patient test adherence will enable faster intervention and treatment, leading to improved patient outcomes and decreased overall diabetes-related healthcare costs—a win for all in the fight against chronic disease.
References
- https://www.cdc.gov/chronicdisease/about/index.htm#:~:text=Many%20chronic%20diseases%20are%20caused. Accessed 1/25/22.
- https://www.cdc.gov/diabetes/basics/quick-facts.html
- American Diabetes Association. Economic costs of diabetes in the U.S. in 2017. Diabetes Care. 2018;41(5):917-28. doi: 10.2337/dci18-0007
- https://www.cdc.gov/kidneydisease/prevention-risk/make-the-connection.html. Accessed 1/25/22.
- https://www.cdc.gov/nchs/fastats/kidney-disease.htm. Accessed 1/25/22.
- https://adr.usrds.org/2020/chronic-kidney-disease/6-healthcare-expenditures-for-persons-with-ckd. Accessed 1/25/22.
- https://www.cdc.gov/kidneydisease/publications-resources/ckd-national-facts.html. Accessed 1/25/22.
- Savoy M, et al. The role of primary care physicians in managing chronic disease. Dela J Public Health. 2017 Mar 22;3(1):86-93. doi: 10.32481/djph.2017.03.012
- Crocker JB, et al. The impact of point-of-care hemoglobin A1c testing on population health-based onsite testing adherence: a primary-care quality improvement study. J Diabetes Sc Technol. 2021;15(3):561-7. doi: 10.1177/1932296820972751
- https://www.uptodate.com/contents/measurements-of-glycemic-control-in-diabetes-mellitus?search=hba1c%20diabetes&source=search_result&selectedTitle=3~150&usage_type=default&display_rank=3. Accessed 1/25/22.
- American Diabetes Association. Glycemic targets: standards of medical care in diabetes—2021. Diabetes Care. 2021 Jan;44(Suppl 1):S73-S84. Available from: https://doi.org/10.2337/dc21-S006. Accessed 1/25/22.
- https://www.cdc.gov/diabetes/pdfs/library/Diabetes-Report-Card-2019-508.pdf. Accessed 12-17-21. Accessed 1/25/22.
- Imai C, et al. Adherence to guideline-recommended HbA1c testing frequency and better outcomes in patients with type 2 diabetes: a 5-year retrospective cohort study in Australian general practice. BMJ Quality & Safety. 2021;30(9):706-14. doi: 10.1136/bmjqs-2020-012026
- Stratton IM, Adler AI, Neil HA, Matthews DR, Manley SE, Cull CA, et al. Association of glycaemia with macrovascular and microvascular complications of type 2 diabetes (UKPDS 35): prospective observational study. BMJ. 2000;321:405-12.
- Rhyu J, Lambrechts S, Han MA, Freeby MJ. Utilizing point-of-care A1c to impact outcomes – can we make it happen in primary care? Curr Opin Endocrinol Diabetes Obes. 2022 Feb 1;29(1):29-33. doi: 10.1097/MED.0000000000000700.
- Schnell, Oliver et al. “Impact of HbA1c Testing at Point of Care on Diabetes Management.” Journal of diabetes science and technology vol. 11,3 (2017): 611-617. doi:10.1177/1932296816678263
- Crocker JB, Lee-Lewandrowsky E, Lewandrowsky N, et al. Implementation of point-of-care testing in an ambulatory practice of an academic medical center. Am J Clin Pathol. 2014;142(5):640-646.
- Al Hayek, Ayman A et al. “Assessment of Patient Satisfaction with On-Site Point-of-Care Hemoglobin A1c Testing: An Observational Study.” Diabetes therapy: research, treatment and education of diabetes and related disorders vol. 12,9 (2021): 2531-2544. doi:10.1007/s13300-021-01126-7
- Rosa, Lorena de Sousa et al. “Cost-Effectiveness of Point-of-Care A1C Tests in a Primary Care Setting.” Frontiers in pharmacology vol. 11 588309. 19 Jan. 2021, doi:10.3389/fphar.2020.588309)
- Chadee, A et al. “Point-of-Care Hemoglobin A1c Testing: A Budget Impact Analysis.” Ontario health technology assessment series vol. 14,9 1-23. 1 Jul. 2014
- Christofides, Elena A, and Niraj Desai. “Optimal Early Diagnosis and Monitoring of Diabetic Kidney Disease in Type 2 Diabetes Mellitus: Addressing the Barriers to Albuminuria Testing.” Journal of primary care & community health vol. 12 (2021): 21501327211003683. doi:10.1177/21501327211003683
- Schultes B, Emmerich S, Kistler AD, Mecheri B, Schnell O, Rudofsky G. Impact of Albumin-to-Creatinine Ratio Point-of-Care Testing on the Diagnosis and Management of Diabetic Kidney Disease. Journal of Diabetes Science and Technology. October 2021. doi:10.1177/19322968211054520
- Currin, S.D., Gondwe, M.S., Mayindi, N.B. et al. Diagnostic accuracy of semiquantitative point of care urine albumin to creatinine ratio and urine dipstick analysis in a primary care resource limited setting in South Africa. BMC Nephrol 22, 103 (2021). https://doi.org/10.1186/s12882-021-02290-5
- Shephard, Anne K. BSc (Hons)*; Shephard, Mark D.S. PhD*; Halls, Heather J. MSc*; Corso, Olivia BSc†; Mathew, Timothy H. MBBS, FRACP† Innovative Use of Point-of-Care Testing for Chronic Kidney Disease Screening, Point of Care: The Journal of Near-Patient Testing & Technology: June 2011 - Volume 10 - Issue 2 - p 98-101 doi: 10.1097/POC.0b013e31821c6bd0
- Harasemiw, Oksana et al. “Impact of point-of-care screening for hypertension, diabetes and progression of chronic kidney disease in rural Manitoba Indigenous communities.” CMAJ: Canadian Medical Association journal = journal de l'Association medicale canadienne vol. 193,28 (2021): E1076-E1084. doi:10.1503/cmaj.201731
- American Diabetes Association Professional Practice Committee et al. “11. Chronic Kidney Disease and Risk Management: Standards of Medical Care in Diabetes-2022.” Diabetes care vol. 45, Supplement_1 (2022): S175-S184. doi:10.2337/dc22-S011
- https://www.ecfr.gov/current/title-42/chapter-IV/subchapter-G/part-493. Accessed 1/25/22.
- Nichols, James H. “Utilizing Point-of-Care Testing to Optimize Patient Care.” EJIFCC vol. 32,2 140-144. 29 Jun. 2021
- Folkerts, Kerstin et al. “Adherence to Chronic Kidney Disease Screening Guidelines Among Patients with Type 2 Diabetes in a US Administrative Claims Database.” Mayo Clinic proceedings vol. 96,4 (2021): 975-986. doi: 10.1016/j.mayocp.2020.07.037
- Alfego, David et al. “Chronic Kidney Disease Testing Among At-Risk Adults in the U.S. Remains Low: Real-World Evidence from a National Laboratory Database.” Diabetes care vol. 44,9 (2021): 2025-2032. doi:10.2337/dc21-0723
- Davidson, Jaime A. “The increasing role of primary care physicians in caring for patients with type 2 diabetes mellitus.” Mayo Clinic proceedings 85,12 Suppl (2010): S3-4. doi:10.4065/mcp.2010.0466
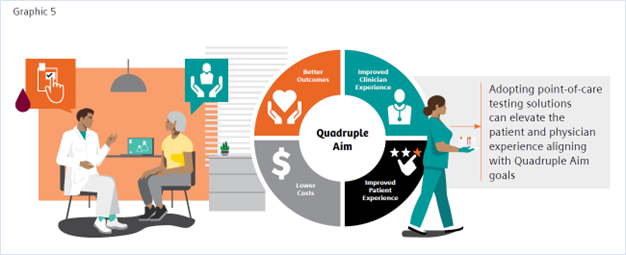
- Bodenheimer, Thomas, and Christine Sinsky. “From triple to quadruple aim: care of the patient requires care of the provider.” Annals of family medicine 12,6 (2014): 573-6. doi:10.1370/afm.1713 (ONLY ADD IF INCLUDED IN GRAPHICS)
