 |
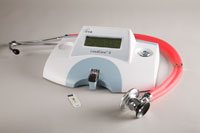
LeadCare II
MAGELLAN BIOSCIENCES
The CDC estimates that lead poisoning still affects 250,000 children in the United States each year. What’s worse, 80% of them suffer from undiagnosed lead poisoning because there are no obvious symptoms, and many children do not receive the lead tests recommended at 1 and 2 years of age. CLIA-waived LeadCare II makes it easy to screen young children, providing lab-accurate results in just three minutes. Because LeadCare II only requires 2 drops of blood, it is the perfect complement to the o
|
CLIA-waived LeadCare® II point-of-care analyzerThe world’s only CLIA-waived blood-lead analyzer, LeadCare II improves clinical outcomes and is ideal for the physicians’ office, health clinic, or outreach-screening program. Safe, fast, easy, and inexpensive to use, ESA developed LeadCare II with a grant from the U.S. Centers for Disease Control. No more waiting days for expensive lab analysis, or worrying about losing track of a child that needs follow-up care. LeadCare II delivers quantitative blood lead results equivalent to those reported by outside laboratories in just three minutes. It improves patient care by providing on-the-spot results, cuts your staffs' result-tracking and administrative time, and qualifies for reimbursement as a quantitative blood lead test (CPT code: 83655).
LeadCare II features:
MAGELLAN BIOSCIENCES
22 Alpha Road | | Chelmsford | MA |
Magellan Biosciences, Inc. was established in October 2004 through the combination of
ESA Biosciences and Dynex Technologies. Magellan's innovative products
include rapid point-of-care systems to screen children and adults for lead poisoning; microbiology products
for blood culture and susceptibility testing that help in the global effort to prevent MRSA and other
drug-resistant infections; as well as automated ELISA processing systems for a wide variety of
immunoassays.

|
|